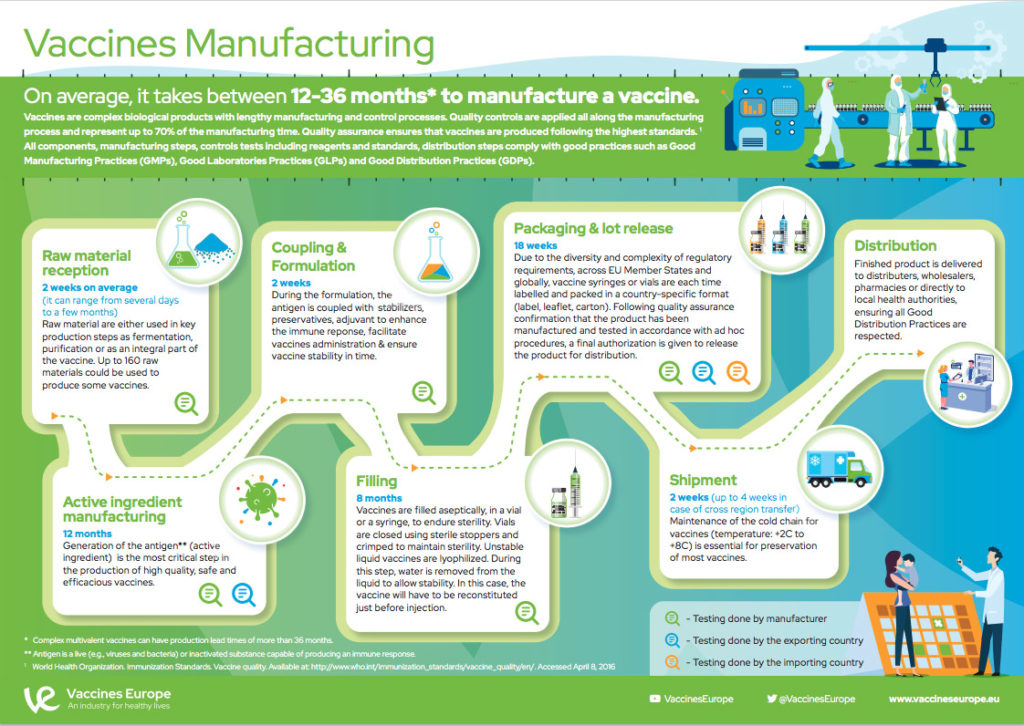
On average, it takes between 12-36 months* to manufacture a vaccine before it is ready for distribution.
Vaccines are complex biological products with lengthy manufacturing and control processes. The quality controls represent up to 70% of the full manufacturing duration.
Successful manufacturing of high-quality vaccines requires international standardization of starting materials, production and quality control testing, and the setting of high expectations for regulatory oversight of the entire manufacturing process from start to finish, all while recognizing that this field is in constant change1.
All the components, manufacturing processes, testing methods, their reagents and standards have to comply with the standards defined for Good Manufacturing Practices (GMP). These strong quality requirements involve ad hoc pharmaceutical quality systems, quality assurance measures and procedures, several quality controls at each stage and an adequate infrastructure and separation of activities to guarantee vaccine identity, purity, sterility, efficacy and safety.
*Complex multivalent vaccines can have production lead times of more than 36 months.
1. World Health Organization. Immunization Standards. Vaccine quality. Available here (Accessed April 8, 2016)
 Members area
Members area

